Clennell, B., Steward, T.G.J., Hanman, K., Needham, T., Benachour, J., Jepson, M., Elley, M., Halford, N., Heesom, K., Shin, E., Molnár, E., Drinkwater, B.W., Whitcomb, D.J., Ultrasound modulates neuronal potassium currents via ionotropic glutamate receptors, Brain Stimulation, 31 Jan 2023.
https://doi.org/10.1016/j.brs.2023.01.1674
Abstract
Backgrond: Focused ultrasound stimulation (FUS) has the potential to provide non-invasive neuromodulation of deep brain regions with unparalleled spatial precision. However, the cellular and molecular consequences of ultrasound stimulation on neurons remains poorly understood. We previously reported that ultrasound stimulation induces increases in neuronal excitability that persist for hours following stimulation in vitro. In the present study we sought to further elucidate the molecular mechanisms by which ultrasound regulates neuronal excitability and synaptic function.
Objective: To determine the effect of ultrasound stimulation on voltage-gated ion channel function and synaptic plasticity.
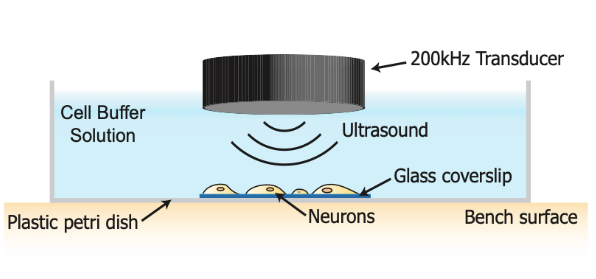
Methods: Primary rat cortical neurons were exposed to a 40 s, 200 kHz pulsed ultrasound stimulus or sham-stimulus. Whole-cell patch clamp electrophysiology, quantitative proteomics and high-resolution confocal microscopy were employed to determine the effects of ultrasound stimulation on molecular regulators of neuronal excitability and synaptic function.
Results: We find that ultrasound exposure elicits sustained but reversible increases in whole-cell potassium currents. In addition, we find that ultrasound exposure activates synaptic signalling cascades that result in marked increases in excitatory synaptic transmission. Finally, we demonstrate the requirement of ionotropic glutamate receptor (AMPAR/NMDAR) activation for ultrasound-induced modulation of neuronal potassium currents.#
Conclusion: These results suggest specific patterns of pulsed ultrasound can induce contemporaneous enhancement of both neuronal excitability and synaptic function, with implications for the application of FUS in experimental and therapeutic settings. Further study is now required to deduce the precise molecular mechanisms through which these changes occur.